FSSAI nutraceutical labelling has emerged as one of the largest make-or-buy considerations for health supplement brands in the rapidly expanding wellness industry of India. Most founders perform a lot of work on formulation, branding and marketing when the complexity of the regulatory side of the label slips through the eye of a needle until one gets notice, rejection or a penalty.
The psychology in this blog is the most common mistakes in labelling, correcting the mistake step by step, the benefits of compliance to any business, and how Foodsure, the food expert consultants and nutraceutical product consultant team, can assist in the process of labelling health supplements and nutraceuticals in a confident way to avoid unlawful errors in their labelling.
Common Psychological Traps:
- Everyone employs these prejudices.
- Mistaken pharma and food regulations.
- Excessive self-confidence in marketing terms.
- Outsourcing compliance to designers, rather than specialists.
Founders tend to believe that nutraceuticals are between food and medicine, since they are in between, especially during nutraceutical food development.
“If it’s safe and effective, the label should be fine.”
Unfortunately, FSSAI nutraceutical labelling does not control intention, but rather evidence and form, even if the nutraceutical food recipe is technically sound.
Why FSSAI Labelling Is Critical for Health Supplements & Nutraceuticals?
This category is strictly regulated by the Food Safety and Standards (Health Supplements, Nutraceuticals, FSMP, Novel food) Regulations.
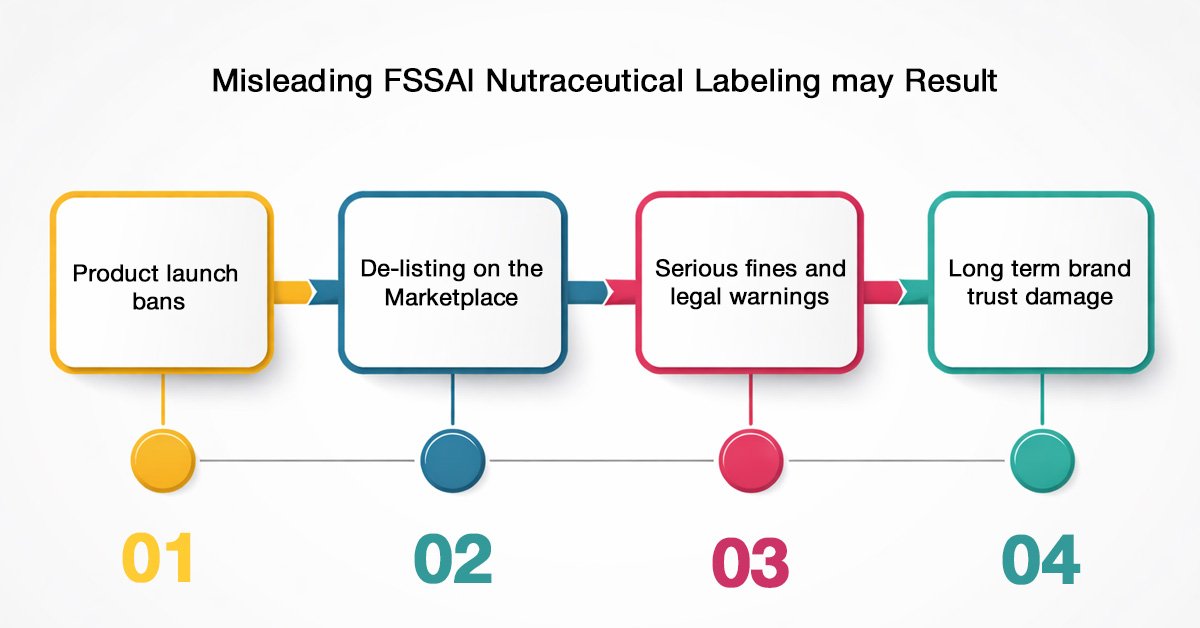
Misleading FSSAI nutraceutical labelling may result in:
- Product launch bans
- De-listing on the marketplace
- Serious fines and legal warnings
- Long-term brand trust damage
This is the reason behind the fact that labelling is not an ultimate activity; it is a plan, integrated with Nutraceutical Product Formulation and compliance strategy.
Mandatory FSSAI Nutraceutical Labelling Requirements
Any health supplement/nutraceutical label should contain:
S. No. | Mandatory Requirement | FSSAI Expectation |
1 | Product Classification | Must clearly state Health Supplement or Nutraceutical as per FSSAI regulations |
2 | Ingredient List | All ingredients are listed in descending order with permitted sources |
3 | Active Ingredient Declaration | Quantity of vitamins, minerals, and botanicals per serving |
| 4 | Nutritional Information Panel | Energy, protein, carbs, fats, and actives as applicable |
| 5 | Recommended Usage & Dosage | Clear daily intake instructions and consumption method |
| 6 | Mandatory Disclaimer | “Not intended to diagnose, treat, cure or prevent any disease” |
| 7 | FSSAI Logo & License Number | Clearly visible and legible on the label |
Common Labelling Problems in Nutraceuticals
Despite obvious rules, brands end up failing because of:
1. Unapproved Health Claims
Claims like:
- “Boosts immunity”
- “Treats diabetes”
- “Cures deficiency”
These go against FSSAI norms without clinical support.
2. Incorrect Dosage or Units
Audit failure can be due to a mismatch of nutraceutical product formulation and label dosage.
3. Pharma-Style Claims
The terms such as therapy, medicine, or treatment are forbidden for any health supplement business product.
4. Missing Warnings
Herbal extracts, caffeine in particular, or vitamins in excess of the RDA limits in a nutraceutical recipe.
How to Correct FSSAI Labelling Issues?
During pre-launch or during the time of sale, corrections should be systematic.
Steps | Corrective Step | Purpose |
1 | Validate Product Category | Prevents misclassification and rejection |
2 | Audit Ingredients & Dosage | Ensures compliance with the FSSAI permitted limits in nutraceutical food formulation |
3 | Review Health Claims | Removes unapproved or risky claims |
| 4 | Update Label Content & Layout | Meets font size, sequence, and readability norms |
| 5 | Verify Disclaimers & Warnings | Ensures mandatory statements are included |
| 6 | Final Compliance Review | Confirms launch-ready, regulator-safe labelling |
Benefits of Compliant FSSAI Nutraceutical Labelling
Compliance brings much less than brands that are labelled correctly, especially for a scaling Health Supplement Business:
- Quick Launches of products: No back-and-forth waiting in regulation.
- Marketplace Approval: Onboarding of e-commerce and retail chains is a smooth journey.
- Consumer Trust: The clear labels represent credibility and safety.
- Long-Term Brand Protection: The chances of penalties, recall programs, or prohibitions are minimised.
- Easier Line Extensions: Once the systems are made compliant, the scaling is more rapid for nutraceutical food development expansion.
Our Role in Nutraceutical Compliance
FSSAI labelling of health supplements and nutraceuticals is not a game of luck; it has to be experienced.
Foodsure, a major team of food consultants and nutraceutical product consultants, is in support of brands in:
- Regulatory Strategy: Proper classification and compliance roadmap since the first day.
- Product and Label Audit: validation of ingredients, checking of dosages and verification of claims.
- Label & Packaging Compliance: Having artwork to be compliant with the FSSAI.
- End-to-End Documentation: F2L license updates and audit.
- Food Product Development Support: It is Compliance-led innovation, and the products are not developed further; they are ready to be launched.
Ready to Get Your Nutraceutical Label Right?
You are either launching a new health supplement, adding to an already existing nutraceutical portfolio or getting FSSAI notices or rejections in your health supplement business.
Foodsure would get you correct in no time.
Register for a free compliance consultation now!
READY TO SCALE YOUR FOOD?
Launch a Nutraceutical Food Product
From development and stability testing to full-scale commercial food product development, Foodsure supports end-to-end manufacturing.
FAQs
Is it different from health supplements and nutraceuticals?
Yes, they are subject to certain regulations of FSSAI with more strict claim, dosage, and disclaimer provisions.
Is nutraceutical labelling obligatory to include a disclaimer?
No, omissions in the disclaimers may result in non-compliance and fines.
What will occur in the case of a non-compliant label?
FSSAI can give notices, fines, stop sale or demand relabeling.
What are the ways food consultants can assist in FSSAI labelling?
To prevent problems with the regulations, the food consultants provide proper classification, claims, dosage, and labelling structure.
When FSSAI labelling: Before or after product development?
Preferably, when developing food products, to prevent the expenses of reformulation and relabelling at a later stage.