India’s wellness market is growing rapidly, driving strong demand for nutraceutical and functional food products such as supplements, protein blends, and immunity boosters. However, before launching any product, businesses must complete proper FSSAI registration and obtain a valid nutraceutical license in India.
Since these products contain concentrated nutrients and health claims, they are regulated under specific FSSAI nutraceutical regulations. To secure an FSSAI license for nutraceuticals, companies must strictly follow the FSSAI guidelines for nutraceuticals, including correct product classification, approved ingredients, lab testing, documentation, and compliance checks.
This guide provides an overview of the key steps involved in obtaining approval smoothly and legally.
Key Regulations For Nutraceutical And Functional Food Products
The Food Safety and Standards Authority of India (FSSAI) looks after food safety nationwide. It sets the standards for all the food products, including nutraceuticals and functional foods, under the Food Safety and Standards Act, 2006. The main FSSAI regulations are:
- FSSAI regulates nutraceuticals and functional foods in India under the Food Safety and Standards Act, 2006.
- Governed by FSSAI Nutra Regulations 2016 (amended 2022/2024).
- Only FSSAI-approved ingredients allowed; new ingredients need safety approval.
- Products must fit categories like Health Supplements, Nutraceuticals, FSMP, Probiotics, and Prebiotics.
- No disease cure claims; only approved health claims with evidence.
- GMP compliance is mandatory.
- Apply via the FoSCoS portal for a nutraceutical license in India.
- Regular FSSAI audits and compliance updates required.
Read more: FSSAI Compliance for Food Startups.
How To Do FSSAI Registration For Nutraceutical And Functional Food Products?
The process of licensing and FSSAI registration for nutraceutical and functional food products is conducted through the Food Safety Compliance System (FoSCoS) portal. The FSSAI registration and licensing process includes:
1. Understand The Categories As A Functional Food Or Nutraceutical
Before FSSAI registration, brands must correctly categorise their product under the Food Safety and Standards Act, 2006, to ensure smooth nutraceutical and functional food compliance in India.
Main FSSAI Categories:
- Functional Foods: Foods offering health benefits beyond basic nutrition.
- Nutraceuticals: Purified or extracted food-based products with physiological benefits (e.g., omega-3, curcumin tablets).
- Health Supplements: Vitamins, minerals, amino acids, botanicals, enzymes, or probiotics intended to supplement the diet under FSSAI health supplement regulations.
Proper product classification is essential for obtaining a nutraceutical license in India.
2. Choose the Correct Type of FSSAI License
Depending on your business size and turnover, you must choose the correct FSSAI license for nutraceuticals and functional foods in India:
- Basic FSSAI Registration: For food businesses with an annual turnover below ₹12 lakh.
- State FSSAI License: For turnover between ₹12 lakh and ₹20 crore or operations within one state.
- Central FSSAI License: For manufacturers, importers, exporters, or businesses with turnover above ₹20 crore or multi-state operations.
For any functional food or nutraceutical product with novel ingredients or health claims, separate FSSAI product approval under the Nutraceutical Division is required, along with the FSSAI license, to ensure full FSSAI compliance in India.
3. Formulate the Food Product As Per The FSSAI Ingredient Guidelines
Food recipe formulation consultants ensure full FSSAI compliance for nutraceuticals and functional foods in India by following strict ingredient regulations:
- Use only FSSAI-approved ingredients and additives listed under Schedule VI of the FSSAI Nutra Regulations 2016.
- Follow maximum permissible limits for vitamins, minerals, amino acids, and bioactives as per FSSAI guidelines.
- Avoid any prohibited substances such as hormones, steroids, or psychotropic compounds to maintain regulatory compliance.
This ensures smooth approval under FSSAI nutraceutical regulations and reduces the risk of license rejection.
4. Conduct Lab And Stability Testing Of Nutraceutical Or Functional Food
FSSAI requires scientific validation and safety testing for all nutraceuticals and functional foods in India before approval. Proper lab testing and stability studies are essential for:
- Microbial contamination testing to ensure product safety.
- Heavy metal analysis to meet FSSAI safety standards.
- Nutritional composition verification as per label claims and FSSAI nutraceutical regulations.
- Shelf-life and stability testing under different storage conditions to support label declarations.
These tests are critical for smooth FSSAI product approval and nutraceutical license compliance in India.
5. Prepare Documentation For FSSAI Food Product Approval
If your nutraceutical or functional food product contains novel ingredients or exceeds permissible limits, it falls under the FSSAI product approval system.
Checklist Of Documents Required For FSSAI Food Product Approval
- Photo ID of the Food Business Operator
- Proof of premises (like a rent agreement or sale deed)
- Company formation documents (Partnership Deed, Certificate of Incorporation)
- List of all products (ingredients and finished goods)
- Food safety management plan
- Layout of your processing unit
- List of machinery and equipment
- Water analysis report (if water is an ingredient)
- Details of food product formulation and manufacturing process (with active ingredients and dosages)
- Scientific evidence of safety and efficacy (published studies, clinical trials)
- Certificate of analysis (COA) from NABL-accredited labs
- Ingredient sourcing and quality documents
- Packaging and labelling details
- Declaration of conformity to FSSAI regulations
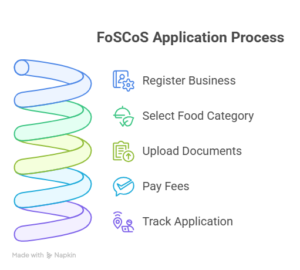
6. Submit Your Application Through the FoSCoS Portal
Once your documents are ready, submit your application on:
- Register your business on https://foscos.fssai.gov.in
- Select food product category- Health supplement, Nutraceutical, Functional food, and more.
- Upload documents and pay the required fees.
- Track your application status online.
Always cross-check your documentation with the latest FSSAI advisories and Q&A circulars. The authority regularly updates norms, particularly for botanicals, probiotics, and dosage limits. Get in touch with the food formulation experts as they help you to remain updated with the new FSSAI rules and regulations.
7. Review And Respond To Queries From The FSSAI Panel
FSSAI may raise queries or ask for clarification during the verification process. These can be:
- Scientific evidence for health claims
- Ingredient safety (ADI, NOAEL, toxicity reports)
- Product stability or manufacturing standards
Have a nutraceutical formulation consultant who is ready to respond to all these queries, as poorly framed responses can delay approvals or result in rejections.
8. Final Approval and License Granting
If everything is perfect:
- You’ll receive a product approval certificate, valid for the approved formulation.
- You can proceed with manufacturing, packaging, and marketing using your FSSAI Central or State License.
- Approved products get a Unique Product Code (UPC), which must be quoted on the label.
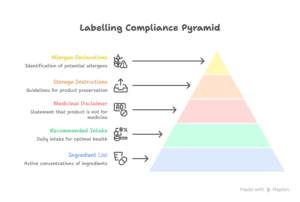
9. Make Sure Of The Labelling Compliance
Labelling is important, especially for functional foods and nutraceuticals. FSSAI mandates the following:
- Ingredient list with active concentrations
- Recommended daily intake (RDI)
- “Not for Medicinal Use” disclaimer
- Storage instructions
- Allergen declarations
10. Post-Market Surveillance And Renewals
After registration, you must:
- Conduct periodic testing of batches.
- Maintain manufacturing and batch records.
- Renew your FSSAI license every 1-5 years, depending on type.
- Stay updated with regulatory amendments.
A quality assurance system (HACCP, GMP) in place increases credibility and ensures continued compliance.
Read More: How Food Consultants Simplify Food Safety Regulations For You
Still Have Queries?
Launching a functional food or nutraceutical in India requires complete FSSAI compliance and proper registration, not just a good product. If you’re unsure about the FSSAI license, documents, or nutraceutical approval process, we can help simplify your functional food registration in India and avoid delays.
At Foodsure, our food formulation consultants don’t just advise, they walk with you from concept to compliance. In one free session, you’ll get answers customised to your product, your category, and your goals. Send us your queries on +91 8130404757
Book your free consultation today!
FAQs
Q1. What products need FSSAI registration under the nutraceutical and functional food rules?
Products like nutraceuticals, functional foods, health supplements, FSDU, probiotics, prebiotics, and novel foods require FSSAI registration.
Q2. Which type of FSSAI license is required for nutraceutical manufacturing?
Depending on turnover and scale, you may require Basic Registration, State License, or Central License.
Q3. What testing is needed for nutraceutical products before FSSAI approval?
Microbial testing, heavy-metal residue testing, nutritional verification, and stability/shelf-life testing are required.
Q4. Are all ingredients allowed in nutraceutical products under FSSAI?
Only ingredients from FSSAI’s approved list are allowed. Novel or unlisted ingredients require special approval.
Q5. Can nutraceutical products claim to cure diseases?
No. FSSAI prohibits disease-cure or therapeutic claims for nutraceutical and functional food products.
Q6. What documents are required for FSSAI nutraceutical licensing?
Documents include formulation details, ingredient list, COA/test reports, premises proof, label drafts, and manufacturing details.
Q7. What are the consequences of non-compliance with FSSAI regulations?
Non-compliance can lead to license rejection, cancellation, product recall, penalties, or restrictions on sale.